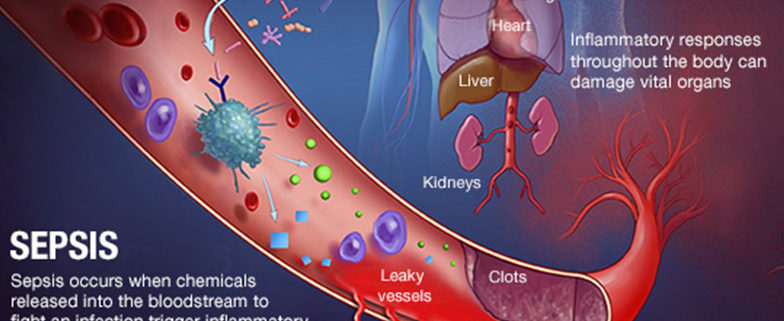
SEPSIS
Sepsis is defined as the presence (probable or documented) of infection together with systemic features of inflammation. Sepsis is identical to SIRS, except that it must result specifically from infection rather than from any of the noninfectious insults that may also cause SIRS (see the image below). That sepsis and SIRS are phenotypically similar underscores a common inflammatory pathway causing both.

Detrimental host responses to infection occupy a continuum that ranges from sepsis to severe sepsis to septic shock and multiple organ dysfunctions syndrome (MODS). The specific clinical features depend on where the patient falls on that continuum. Patients with sepsis may present in a myriad of ways, and a high index of clinical suspicion is necessary to identify subtle presentations.

Symptoms of sepsis include:
- a fever above 101ºF (38ºC) or a temperature below 96.8ºF (36ºC)
- heart rate higher than 90 beats per minute
- respiratory rate higher than 20 breaths/min or arterial carbon dioxide tension below 32 mm Hg
- white blood cell (WBC) count higher than 12,000/µL, lower than 4000/µL, or including more than 10% bands
Two of these symptoms are required to make a diagnosis of sepsis.
SEVERE SEPSIS
Severe sepsis is sepsis-induced tissue hypoperfusion or organ dysfunction/failure. One or more of the following signs are required to establish a diagnosis of severe sepsis:
- patches of discolored skin
- decreased urination
- changes in mental ability
- low platelet count
- problems breathing
- abnormal heart functions
- chills due to fall in body temperature
- unconsciousness
- extreme weakness
Sepsis can quickly become overwhelming; as sepsis progresses, the blood pressure drops dangerously low, and organs can stop functioning correctly. This can take hours or days, depending on the individual.
SEPTIC SHOCK
Septic shock occurs in a subset of patients with sepsis and comprises an underlying circulatory and cellular/metabolic abnormality that is associated with a 50 percent mortality rate.
Symptoms of septic shock include the symptoms of severe sepsis, plus persisting hypotension that requires vasopressors to maintain a mean arterial pressure of 65-mm Hg or higher and a serum lactate level greater than 2-mmol/L (18-mg/dL) despite adequate volume resuscitation.
PATHOPHYSIOLOGY OF SEPSIS
Sepsis has been referred to as a process of malignant intravascular inflammation. Normally, a potent, complex, immunologic cascade ensures a prompt protective response to microorganism invasion in humans. A deficient immunologic defense may allow the infection to become established; however, an excessive or poorly regulated response may harm the host through a maladaptive release of indigenously generated inflammatory compounds. (see the image below).
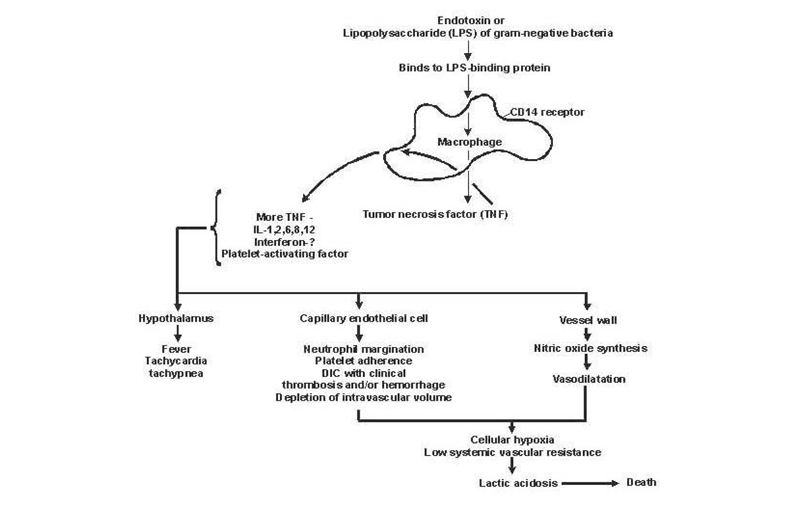
Lipid A and other bacterial products release cytokines and other immune modulators that mediate the clinical manifestations of sepsis. Interleukins, tumor necrosis factor (TNF)-α, interferon gamma (IFN-γ), and other colony-stimulating factors are produced rapidly within minutes or hours after interactions of monocytes and macrophages with lipid A.
Inflammatory mediator release becomes a self-stimulating process, and release of other such mediators, including interleukin (IL)-1, platelet activating factor, IL-2, IL-6, IL-8, IL-10, and nitric oxide (NO), further increases cytokine levels. This leads tocontinued activation of polymorphonuclear leukocytes (PMNs), macrophages, and lymphocytes; proinflammatory mediators recruit more of these cells. All these processes create a state of destructive immunologic dissonance.
Sepsis is described as an auto-destructive process that permits extension of the normal pathophysiologic response to infection to involve otherwise normal tissues and results in MODS. Organ dysfunction or organ failure may be the first clinical sign of sepsis, and no organ system is immune from the consequences of the inflammatory excesses of sepsis. Mortality increases as organ failure increases.
Septic shock and SIRS are characterized by reversible myocardial depression, which can prove resistant to catecholamine and fluid administration. Circulating “myocardial depressant factor”—probably representing the synergistic effects of TNF-α, IL-1β, other cytokines, and NO—is implicated in pathogenesis. The two characteristics of this acute stress myocardial depression are impaired adrenergic responsiveness and diastolic dysfunction leading to relative catecholamine resistance and small rather than dilated hearts. Macrovascular myocardial ischemia and hypoperfusion are unlikely contributors.
In severe sepsis and septic shock, microcirculatory dysfunction and mitochondrial depression cause regional tissue distress, and regional dysoxia therefore persists. This condition is termed microcirculatory and mitochondrial distress syndrome (MMDS). Sepsis-induced inflammatory autoregulatory dysfunction persists, and oxygen need is not matched by supply, leading to MODS.
Redistribution of intravascular fluid volume, resulting from a reduced arterial vascular tone, diminished venous return from venous dilation, and release of myocardial depressant substances causes hypotension.
Cytokines are responsible for the clinically observable effects of bacteremia in the host. Impaired pulmonary, hepatic, or renal function may result from excessive cytokine release during the septic process.
Sepsis can affect multiple organs or the entire body, even without septicemia.
EXTRACORPOREAL REMOVAL OF TOXINS
One cannot discard the role of endotoxin in the pathophysiology of Gram-negative sepsis, but antiendotoxin strategies have not been particularly effective. Moreover, endotoxin is not released only in Gram-negative infections; indeed, measuring endotoxin levels is not very helpful to distinguish Gram-negative from Gram-positive infections.
SIGNS OF END-ORGAN HYPOPERFUSION
These signs include the following:
- Warm, flushed skin may be present in the early phases of sepsis. The skin may become cool and clammy with progression to shock due to redirection of blood flow to core organs. Decreased capillary refill, purpura cyanosis, or mottling may be seen.
- Altered mental status, obtundation, restlessness
- Oliguria or anuria due to hypoperfusion
- Ileus or absent bowel sounds
SEPSIS-INDUCED MYOCARDIAL DYSFUNCTION (SIMD)
Sepsis-induced myocardial dysfunction (SIMD) is one of the major predictors of morbidity and mortality of sepsis.
Infection dials up the body’s immune response, generating systemic inflammation that can cause plaque to rupture and create blockages that lead to heart attack and stroke. Patients tend to have high levels of inflammatory markers circulating in their blood long after recovering from an infection.
Myocardial dysfunction is one of the main predictors of poor outcome in septic patients, with mortality rates next to 70%. During sepsis-induced myocardial dysfunction, both ventricles can dilate and diminish its ejection fraction, having less response to fluid resuscitation and catecholamines. The degree of SIMD is very variable, ranging from mild to severe. Sometimes the degree of myocardial depression may be so severe that it imitates a cardiogenic shock but typically is assumed to be reversible within 7-10 days.
With the introduction of hemodynamic monitoring with pulmonary artery catheter, the existence of myocardial depression in patients with septic shock was confirmed, showing depressed cardiac response to volume infusion.
TROPONIN
Troponin is a sensitive marker of myocardial injury, and ‘non-coronary’ troponin elevation is common among critically ill patients. Elevated troponin levels are observed in 43% across all intensive care patient groups. The estimated prevalence of positive troponin in the context of sepsis is 61%.
The mechanism of myocyte insult in severe sepsis and septic shock, in the absence of thrombotic acute coronary syndrome, leading to elevated serum troponin, is not yet fully understood. Myocardial depressive factors (inflammatory mediators, endotoxins), microvascular dysfunction, and increased myocardial cell membrane permeability in conjunction with myocardial oxygen demand–supply mismatch, are potential explanations for sepsis-induced troponin elevation. In this setting, troponin elevation occurs in the absence of myocyte necrosis, and this hypothesis is supported by clinical observations that myocardial depression in the context of sepsis is a reversible process in most surviving patients.
ELECTROCARDIOGRAPH IN SIMD
SIMD can be accompanied by specific changes in the electrocardiogram (ECG), which can be very similar to those occurring during acute coronary syndromes, including ST segment depression or elevation, Q wave appearance, new left bundle branch block, peaked T wave, lengthening of the QTc interval or positive J waves (Osborn wave) appearance. The ECG may suffer temporary modifications, with an initial predominance of ST segment elevation followed by the appearance of a Q wave. These ECG changes may disappear with time. Interestingly, the ECG may suggest the diagnosis of myocardial dysfunction.
CENTRAL NERVOUS SYSTEM DYSFUNCTION
Involvement of the central nervous system (CNS) in sepsis produces encephalopathy and peripheral neuropathy. The pathogenesis is poorly defined but is probably related to systemic hypotension, which can lead to brain hypoperfusion.
MULTIPLE ORGAN DYSFUNCTION IN SEPSIS
Multiple organ dysfunction syndrome (MODS) is defined as a clinical syndrome characterized by the development of progressive and potentially reversible physiologic dysfunction in 2 or more organs or organ systems that is induced by a variety of acute insults, including sepsis.
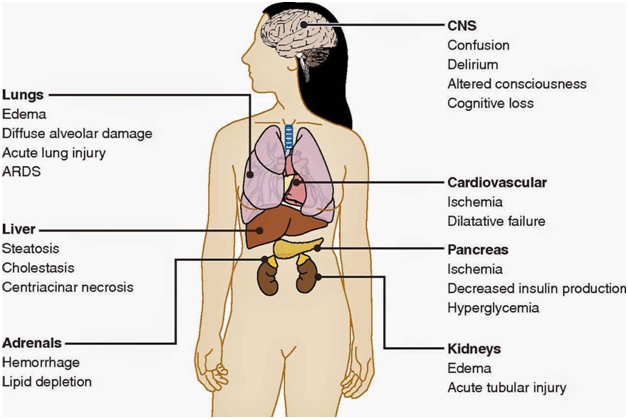
In the more common form of MODS, the lungs are the predominant, and often the only organ system affected until very late in the disease. Patients with this form of MODS most often present with a primary pulmonary disorder (e.g., pneumonia, aspiration, lung contusion, near-drowning, chronic obstructive pulmonary disease exacerbation, hemorrhage, or pulmonary embolism).
Lung disease progresses to meet ARDS criteria. Encephalopathy or mild coagulopathy may accompany pulmonary dysfunction, which persists for 2-3 weeks. At this time, the patient either begins to recover or progresses to fulminant dysfunction in another organ system. Once another major organ dysfunction occurs (see Table 1 below), these patients frequently do not survive.
Table. Criteria for Organ Dysfunction (Open Table in a new window)
| Organ System | Mild Criteria | Severe Criteria |
| Pulmonary | Hypoxia or hypercarbia necessitating assisted ventilation for 3-5 days | ARDS requiring PEEP >10 cm H2 O and FI O2< 0.5 |
| Hepatic | Bilirubin 2-3 mg/dL or other liver function tests >2 × normal, PT elevated to 2 × normal | Jaundice with bilirubin 8-10 mg/dL |
| Renal | Oliguria (< 500 mL/day) or increasing creatinine (2-3 mg/dL) | Dialysis |
| Gastrointestinal | Intolerance of gastric feeding for more than 5 days | Stress ulceration with need for transfusion, acalculous cholecystitis |
| Hematologic | aPTT>125% of normal, platelets < 50-80,000 | DIC |
| Cardiovascular | Decreased ejection fraction with persistent capillary leak | Hyperdynamic state not responsive to pressors |
| CNS | Confusion | Coma |
| Peripheral nervous system | Mild sensory neuropathy | Combined motor and sensory deficit |
| aPTT = activated partial thromboplastin time; ARDS = acute respiratory distress syndrome; CNS = central nervous system; DIC = disseminated intravascular coagulation; FI O2 = fraction of inspired oxygen; PEEP = positive end-expiratory pressure; PT = prothrombin time. |
In the second, less common, form of MODS, the presentation is quite different. Patients affected by this form often have an inciting source of sepsis in organs other than the lung; the most common sources are intra-abdominal sepsis, extensive blood loss, pancreatitis, and vascular catastrophes. ALI or ARDS develops early, and dysfunction in other organ systems (hepatic, hematologic, cardiovascular, and renal) also develops much sooner than in the first form of MODS. Patients remain in a pattern of compensated dysfunction for several weeks, at which time they either recover or deteriorate further and die.
Tachypnea is a common and often underappreciated feature of sepsis. It is an indicator of pulmonary dysfunction and is commonly found in pneumonia and ARDS, both of which are associated with increased mortality in sepsis.
The frequency of ARDS in sepsis has been reported to range from 18% to 38% (with gram-negative sepsis, from 18% to 25%). The severity of ARDS may range from mild lung injury to severe respiratory failure. The onset of ARDS usually is within 12-48 hours of the inciting event. The patients demonstrate severe dyspnea at rest, tachypnea, and hypoxemia; anxiety and agitation are also present.
Chest radiography detects infiltrates in about 5% of febrile adults without localizing signs of infection; accordingly, it should be routine in adults who are febrile without localizing symptoms or signs and in patients who are febrile with neutropenia and without pulmonary symptoms. Chest radiography is useful in detecting radiographic evidence of ARDS, which carries a high mortality. The discovery of such evidence on a chest x-ray should prompt consideration of early intubation and mechanical ventilation, even if the patient has not yet shown signs of overt respiratory distress.
In adult patients with suspected intra-abdominal infection who are not undergoing immediate laparotomy, CT scanning of the abdomen is preferable to abdominal radiography. CT is the imaging modality of choice for excluding an intra-abdominal abscess or a retroperitoneal source of infection. Obesity or the presence of excessive intestinal gas markedly interferes with abdominal imaging by ultrasonography; therefore, CT is preferred in this setting.
LABORATORY STUDIES
A complete blood cell (CBC) count with differential should be obtained. An adequate hemoglobin concentration is necessary to ensure oxygen delivery in shock; hemoglobin should be maintained at a level of 8 g/dL.
Acute-phase reactants and platelets usually increase at the onset of any serious stress. With persistent sepsis, the platelet count will fall, and disseminated intravascular coagulation (DIC) may develop.
The white blood cell (WBC) differential and the WBC count may predict the existence of a bacterial infection. In adults who are febrile, a WBC count higher than 15,000/µL or a neutrophil band count higher than 1500/µL is associated with a high likelihood of bacterial infection.
A metabolic assessment should be performed with measurement of serum electrolytes, including magnesium, calcium, phosphate, and glucose, at regular intervals. Renal and hepatic function should be assessed with measurement of serum creatinine, blood urea nitrogen (BUN), bilirubin, alkaline phosphate, and alanine aminotransferase (ALT).
Arterial blood gas testing is indicated.
Measurement of serum lactate provides an assessment of tissue hypoperfusion. Elevated serum lactate indicates that significant tissue hypoperfusion exists with the shift from aerobic to anaerobic metabolism. This signals a worse degree of shock and a higher mortality.
Coagulation status should by assessed by measuring the prothrombin time (PT) and the activated partial thromboplastin time (aPTT). Patients with clinical evidence of coagulopathy require additional tests to detect the presence of DIC.
Although indiscriminate use of blood cultures has low utility, blood culture is the primary modality for facilitating the diagnosis of intravascular infections (e.g., endocarditis) and infections of indwelling intravascular devices. Two populations—people who abuse intravenous (IV) drugs and patients with prosthetic heart valves—are at high risk for endocarditis.
Patients at risk for bacteremia include adults who are febrile with elevated WBC or neutrophil band counts, elderly patients who are febrile, and patients who are febrile and neutropenic. These populations have a 20-30% incidence of bacteremia. The incidence of bacteremia is at least 50% in patients with sepsis and evidence of end-organ dysfunction.
A urinalysis and a urine culture should be ordered for every patient who is in a septic state. Urinary infection is a common source of sepsis, especially in elderly individuals. Adults who are febrile without localizing symptoms or signs have a 10-15% incidence of occult urinary tract infection (UTI).
Secretions or tissue for Gram stain and culture should be obtained from sites of potential infection. Generally, the Gram stain is the only available test for immediately documenting the presence of bacterial infection and guiding the choice of initial antibiotic therapy.
UNIQUE LABORATORY FINDINGS
Laboratory and clinical features that may suggest an underlying etiology of sepsis are as follows:
- Leukocytosis (WBC count >12,000/µL) or leukopenia (WBC count < 4000/µL)
- Normal WBC count with greater than 10% immature forms (left shift with bandemia)
- Hyperglycemia (plasma glucose level >140 mg/dL or 7.7 mmol/L) in the absence of diabetes
- Plasma C-reactive protein level of more than two standard deviations above the reference value
- Arterial hypoxemia (PaO2/FiO2 ratio < 300 mm Hg)
- Acute oliguria (urine output < 0.5 mL/kg/hour for at least 2 hours despite adequate fluid resuscitation)
- Creatinine increase > 0.5 mg/dL or 44.2 mmol/L
- Coagulation abnormalities (INR >1.5 or PTT >60 seconds)
- Thrombocytopenia (platelet count < 100,000/µL)
- Hyperbilirubinemia (plasma total bilirubin > 4 mg/dL or 70 mmol/L)
- Adrenal insufficiency (e.g., hyponatremia, hyperkalemia) and euthyroid sick syndrome can also be found in sepsis.
- Hyperlactatemia (serum lactate >2 mmol/L) can result from organ hypoperfusion in the presence or absence of hypotension and indicates a poor prognosis. A serum lactate level of 4 mmol/L or more (especially arterial lactate) indicates septic shock.
- Plasma procalcitonin and presepsin elevation is associated with bacterial infection and sepsis.
HEMATOLOGICAL
Severe sepsis or septic shock can cause small blood clots to form throughout the body. These clots can block the flow of blood and oxygen to vital organs and other parts of the body. This increases the risk of organ failure and tissue death (gangrene).
If hemoglobin levels fall below 7g/dL, red blood cell transfusion is recommended to a target hemoglobin range of 7-9 g/dL. Even in the absence of apparent bleeding, patients with severe sepsis should receive platelet transfusion if platelet counts fall below 10 × 109/L (10,000/µL). Disseminated intravascular coagulopathy should first be ruled out with fibrinogen split products and peripheral smears. Platelet transfusion may also be considered when bleeding risk is increased, and platelet counts are below 20 × 109/L (20,000/µL). Patients who are to undergo surgery or other invasive procedures may require higher platelet counts (e.g., ≥50 × 109/L [50,000/µL]).
As the treatment of septic shock involves multiple diverse interventions, all of which need to be performed rapidly without delay, a team approach should be used, not only during working hours but 24-h a day.
COAGULOPATHY
Subclinical coagulopathy, signaled by a mild elevation of the thrombin time (TT) or activated partial thromboplastin time (aPTT) or a moderate reduction in the platelet count, is extremely common; however, overt disseminated intravascular coagulation (DIC) may also develop. Protease-activated receptors (PARs), especially PAR 1, form the molecular link between coagulation and inflammation; PAR1 exerts cytoprotective effects when stimulated by activated protein C or low-dose thrombin but exerts disruptive effects on endothelial-cell barrier function when activated by high-dose thrombin.
LACTATE LEVELS
Septic shock is always associated with increased blood lactate levels, and blood lactate levels should be measured serially (typically every hour) to be sure they are decreasing with time, which indicates that the resuscitation process is effective. If lactate levels do not decrease, then the diagnosis or ongoing treatment should be reexamined.
Lactic acidosis of septic shock usually causes anion gap metabolic acidosis. Administration of bicarbonate has the potential to worsen intracellular acidosis. Correction of acidemia with sodium bicarbonate has not been proved to improve hemodynamics in critically ill patients with increased blood lactate levels. Nevertheless, bicarbonate therapy has been used in cases where the pH is less than 7.20 or the bicarbonate level is lower than 9 mmol/L, though no data to support this practice exist.
IMAGING STUDIES
In patients with severe sepsis, a chest radiograph should be obtained because the clinical examination is unreliable for diagnosing Pneumonia. Clinically occult infiltrates have been detected by routine use of chest radiography in adults who are febrile without localizing symptoms or signs and in patients who are febrile and neutropenic without pulmonary symptoms. Supine and upright or lateral decubitus abdominal films may be useful when an intra-abdominal source is suspected.
Ultrasonography is the imaging modality of choice when a biliary tract infection is suspected of being the source of sepsis.
Computed tomography (CT) is the imaging modality of choice for excluding an intra-abdominal abscess or a retroperitoneal source of infection. A CT scan of the head should be obtained when there is evidence of increased intracranial pressure (papilledema), when factors suggesting focal mass lesions (e.g., focal defects, previous sinusitis or otitis, recent intracranial surgery) are present, or before lumbar puncture (LP) when meningitis is suspected. MRI scans can identify soft tissue infections.
When clinical evidence of a deep soft tissue infection exists (e.g., crepitus, bullae, hemorrhage, or a foul-smelling exudate), a plain radiograph should be obtained. The presence of soft tissue gas and the spread of infection beyond the clinically detectable disease may necessitate surgical exploration.
OTHER DIAGNOSTIC AND SUPPORTIVE PROCEDURES
When meningitis or encephalitis is suspected, LP must be performed on an urgent basis. In patients with an acute fulminant presentation, rapid onset of septic shock, and severe impairment of mental status, bacterial meningitis must be ruled out by means of LP.
Procedures such as cardiac monitoring, noninvasive blood pressure monitoring, and pulse oximetry are necessary because patients often require admission to the intensive care unit (ICU) for invasive monitoring and support. Supplemental oxygen is provided during initial stabilization and resuscitation.
In all patients in septic shock, adequate venous access for volume resuscitation is necessary. A central venous line can also be used to monitor central venous pressure for assessment of intravascular volume status.
An indwelling urinary catheter used to monitor urinary output can serve as a marker for adequate renal perfusion and cardiac output.
Patients in whom septic shock associated with acute lung injury or right-sided heart failure require either right-heart catheterization with a pulmonary artery (Swan-Ganz) catheter or a transpulmonary thermodilution device (e.g., PiCCO, Vigileo) to guide therapy. These catheters provide an assessment of the volume status of a patient who is in a septic state. Cardiac output measurements can be obtained. Furthermore, determination of mixed venous oxygenation from the pulmonary artery catheter is helpful in determining the status of tissue oxygenation.
Dynamic hemodynamic monitoring devices using pulse pressure and stroke volume variation are used in some centers when the patients are in sinus rhythm and on mechanical ventilation without spontaneous breathing to define volume responsiveness and assess dynamic arterial tone, both useful in deciding on resuscitation treatment options.
Most patients who are in a septic state experience respiratory distress secondary to severe sepsis or as a manifestation of septic shock. Pulmonary dysfunction of sepsis (i.e., acute respiratory distress syndrome [ARDS]) also may occur. These patients need intubation and mechanical ventilation for optimum respiratory support.
SURGICAL DRAINAGE AND DEBRIDEMENT
Patients with infected foci should be taken for definitive surgical treatment after initial resuscitation and administration of antibiotics. When an infected focus persists, there is little to be gained from spending hours on attempting to stabilize the patient.
Infectious processes require expeditious surgical drainage or debridement for source control, even if the patient does not appear stable. Without emergency surgical treatment, the patient’s condition may not improve.
CDC’S RECOMMENDATION
The CDC recommends that family members of susceptible patients should be advised of the common symptoms of sepsis:
- a fever above 101ºF (38ºC) or a temperature below 96.8ºF (36ºC)
- heart rate higher than 90 beats per minute
- breathing rate higher than 20 breaths per minute
- probable or confirmed infection
- skin changes
- confusion, among other things
Two of these symptoms must be present for a diagnosis of sepsis. There’s no one symptom of sepsis, but rather it has a combination of symptoms.
HISTORY
Symptoms of sepsis are usually nonspecific and include fever, chills, and constitutional symptoms of fatigue, malaise, anxiety, or confusion. These symptoms are not pathognomonic for infection and may also be observed in a wide variety of noninfectious inflammatory conditions. In addition, they may be absent in patients with serious infections, especially in elderly individuals.
Because systemic inflammatory response syndrome (SIRS), sepsis, septic shock, and multiple organ dysfunction syndrome (MODS) represent a clinical continuum, the specific features exhibited in any given case depend on where the patient falls on that continuum.
Fever is a common feature of sepsis. Fever of infectious origin results from resetting of the hypothalamus, so that heat production and heat loss are balanced to maintain a higher temperature. The abrupt onset of fever usually is associated with a large infectious load.
Chills are a secondary symptom associated with fever and result from increased muscular activity in an attempt to produce heat and thereby raise the body temperature to the level required to reset the hypothalamus.
Sweating occurs when the hypothalamus returns to its normal set point and senses that the body temperature is above the desired level. Perspiration is stimulated to offload excess body heat through evaporative cooling.
Altered mental function is often observed. Mild disorientation or confusion is especially common in elderly individuals. More severe manifestations include apprehension, anxiety, and agitation, and in some cases, coma may eventually ensue. The mechanism by which mental function is altered is not known, but altered amino acid metabolism has been proposed as a cause of metabolic encephalopathy.
Hyperventilation with respiratory alkalosis is a common feature of sepsis. Stimulation of the medullary ventilatory center by endotoxins and other inflammatory mediators has been proposed as the cause of hyperventilation.
The following localizing symptoms are some of the most useful clues to the etiology of both fever and sepsis:
- Head and neck infections – Earache, sore throat, sinus pain, or swollen lymph glands
- Chest and pulmonary infections – Cough (especially if productive), pleuritic chest pain, and dyspnea
- Abdominal and gastrointestinal (GI) infections – Abdominal pain, nausea, vomiting, and diarrhea
- Pelvic and genitourinary (GU) infections – Pelvic or flank pain, vaginal or urethral discharge, frequency, urgency
- Bone and soft tissue infections – Focal pain or tenderness, focal erythema, edema
PHYSICAL EXAMINATION
The physical examination focuses first on the general condition of the patient. Assess the patient’s overall hemodynamic condition to search for signs of hypoperfusion. Look for signs suggestive of a focal infection. An acutely ill, toxic appearance is a common feature in patients with serious infections.
The vital signs may suggest sepsis, even if fever is absent. As noted, tachypnea is common; tachycardia with an increased pulse pressure also is common.
Measure the body temperature accurately. Because oral temperatures are often unreliable, rectal temperatures should be obtained.
Investigate signs of systemic tissue perfusion. In the early stages of sepsis, cardiac output is well maintained or even increased. Along with the effects of vasodilatory mediators, this may result in warm skin, warm extremities, and normal capillary refill. As sepsis progresses, stroke volume and cardiac output fall. Patients begin to manifest signs of poor distal perfusion, including cool skin, cool extremities, and delayed capillary refill.
The following physical signs suggest focal, usually bacterial, infection:
- Central nervous system (CNS) infection – Profound depression in mental status and meningismus
- Head and neck infections – Inflamed or swollen tympanic membranes, sinus tenderness, pharyngeal exudates, stridor, cervical lymphadenopathy
- Chest and pulmonary infections – Localized rales or evidence of consolidation
- Cardiac infections – Regurgitant valvular murmur
- Abdominal and GI infections – Focal tenderness, guarding or rebound, rectal tenderness, or swelling
- Pelvic and GU infections – Costovertebral angle tenderness, pelvic tenderness, cervical motion pain, and adnexal tenderness
- Bone and soft tissue infections – Focal erythema, edema, infusion, and tenderness
- Skin infections – Petechiae and purpura
SKIN COLOR & TEMPERATURE
It is important to pay attention to the patient’s skin color and temperature. Pallor or grayish or mottled skin are signs of poor tissue perfusion seen in septic shock. In the early stages of sepsis, cardiac output is well-maintained or even increased. The vasodilation may result in warm skin, warm extremities, and normal capillary refill (warm shock). As sepsis progresses, stroke volume and cardiac output fall. The patient begins to manifest the signs of poor perfusion: cool skin, cool extremities, and delayed capillary refill (cold shock). The skin of patients with sepsis may be warm or cold, depending on the adequacy of organ and skin perfusion.
While fever is an important sign of serious infections, the absence of fever could be a significant sign of severe sepsis and septic shock. Hypothermia is more predictive of illness severity and death. Fever may be absent in elderly or immunosuppressed patients.
The hypothalamus resets in sepsis. An inquiry should be made about fever onset (abrupt or gradual), duration, and maximal temperature.
RECOGNITION OF SEPSIS
Sepsis is a medical emergency, and rapid treatment can make all the difference in whether or not a person recovers. If sepsis is at all suspected, the patient should be brought to medical attention as quickly as possible. Every minute and hour counts because the infection can spread quickly.
Nursing staffs often miss early signs of infection, which can start with fever and an elevated heart rate, altered mental status, or not eating. When those symptoms occur, nurses should call a doctor.
Sepsis is treatable if it is identified and treated quickly, and in most cases, leads to a full recovery with no lasting problems.
MANAGEMENT OF SEPTIC SHOCK
Sepsis can quickly progress to septic shock and death if it’s left untreated.
The management of septic shock includes three major components, as shown in Figure 1. Importantly, infection control and hemodynamic support need to be performed promptly and together.
Source control, when necessary, must be accomplished rapidly. Source control can range from relatively simple catheter removal to draining a pus-filled abscess or removing infected tissue.
The likely source of the infection should be determined, and intravenous (IV) empiric antimicrobial agents should be administered until culture results become available, at which point more narrow-spectrum agents can be used.
In addition, therapy aimed at maintaining organ perfusion and respiratory support should be provided when necessary. Dialysis might be necessary if the kidneys are affected. Kidneys help filter harmful wastes, salt, and excess water from the blood. In dialysis, a machine performs these functions.
Various medications are used to treat sepsis, including:
- antibiotics via IV to fight infection
- vasoactive medications to increase blood pressure
- insulin to stabilize blood sugar
- corticosteroids to reduce inflammation
- painkillers
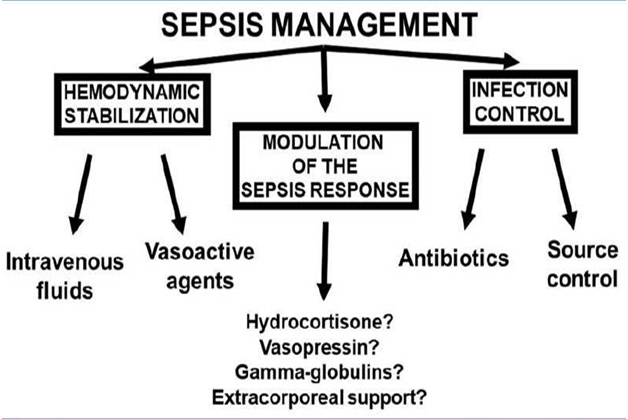
Patients with persisting hypotension requiring vasopressors to maintain mean arterial pressure of more than 65-mm Hg and a serum lactate level of more than 2-mmol/l (18-mg/dl) despite adequate volume resuscitation are said to have septic shock.
The management of sepsis includes early treatment, with fluids and vasopressor agents given simultaneously to assure rapid restoration of an adequate perfusion pressure to limit the development and worsening of organ dysfunction.
Crystalloids are considered as the initial fluid of choice, although albumin can have a place early in patients who are already edematous in a context of hypoalbuminemia (patients with a decompensated cirrhosis represent a typical example), and later if the patient has already received large amounts of crystalloids. Saline solution can be selected initially in the absence of severe acidosis, but chloride levels must be monitored because hyperchloremia can have deleterious effects, most notably on the kidneys. Otherwise, balanced solutions (Ringer’s lactate or PlasmaLyte R) represent the best option.
Administration of crystalloid solution is titrated to a goal of adequate tissue perfusion. CVP should not be used to target resuscitation; it should be used as a stopping rule. If during fluid resuscitation, CVP rapidly increases by more than 2-mm Hg, absolute CVP is greater than 8-12 mm Hg, or signs of volume overload (dyspnea, pulmonary rales, or pulmonary edema on the chest x-ray) occur, fluid infusion as primary therapy needs to be stopped. Patients with septic shock often require a total of 4-6 L or more of a crystalloid solution. However, CVP measurement should not be entirely relied upon because it does not correlate with intravascular volume status or cardiac volume responsiveness.
VASOACTIVE AGENTS
Norepinephrine is the vasopressor of choice; dopamine should no longer be used in this setting. Epinephrine is also best avoided because it is more likely to induce arrhythmias, can reduce splanchnic blood flow, and may increase blood lactate levels as a result of increased cellular metabolism.
GLUCOCORTICOIDS
Recent results have shed new light on the ongoing debate regarding corticosteroid use in sepsis. In the presence of severe septic shock, administration of moderate doses of hydrocortisone (200 mg/day) should be considered until shock is resolved, because relative adrenal insufficiency may develop.
ACTH stimulation test is not recommended for identifying the subset of patients with septic shock or ARDS who should receive corticosteroid therapy. The American College of Critical Care Medicine does not recommend the routine use of free cortisol measurements in critically ill patients. No clear parameters for the normal range of free cortisol in such patients are recognized, and the free cortisol assay is not widely available, despite its advantages over the total serum cortisol assay.
VASOPRESSIN
Vasopressin should not be considered as just another vasopressor agent, but as a form of hormonal support, because vasopressin stores may be decreased in septic shock. Vasopressin derivatives may perhaps decrease edema formation but carry the risk of inducing severe vasoconstriction and decreasing blood flow to the cutaneous, splanchnic, and coronary regions. If vasopressin is used, it should be administered in small doses of about 0.03 U/min without titration, and only in hyperkinetic states demonstrated by the presence of high cardiac output.
GAMMA GLOBULINS
Although there is no place for routine administration of gamma globulins in patients with sepsis, a recent clinical trial suggested a benefit of an IgM-enriched mixture in patients with severe community-acquired pneumonia who had low IgM levels and significant inflammatory response.
PROGNOSIS
Sepsis is a common cause of morbidity and mortality worldwide. The prognosis depends on underlying health status and host defenses, the site and the type of infection, prompt and adequate surgical drainage of abscesses, relief of any obstruction of the intestinal or urinary tract, and appropriate and early empiric antimicrobial therapy.
Failure to attempt aggressive restoration of perfusion early may also be associated with increased mortality risk. A severely elevated lactate level (>4 mmol/L) is associated with a poor prognosis in patients with sepsis.
The prognosis of sepsis treated in a timely manner and with appropriate therapy is usually good. Having a case of severe sepsis increases the risk of a future infection.
POST-SEPSIS SYNDROME
Recovery from sepsis depends on the severity of the condition and any preexisting conditions. Many people who survive will recover completely. However, others will report lasting effects.
Approximately 50 percent of sepsis survivors are affected by post-sepsis syndrome (PSS). This condition includes long-term effects such as:
- damaged organs
- insomnia
- nightmares
- disabling muscle and joint pains
- fatigue
- poor concentration
- lowered cognitive functioning
- lowered self-esteem