Cauda Equina and Conus Medullaris Syndromes
The spinal cord, which is the downward continuation of the medulla that starts just below the foramen magnum, serves as a conduit for the ascending and descending fiber tracts that connect the peripheral and spinal nerves to the brain. The spinal cord projects 31-pairs of spinal nerves to either side (8-cervical, 12-thoracic, 5-lumbar, 5-sacral, 1-coccygeal) that are connected to peripheral nerves.
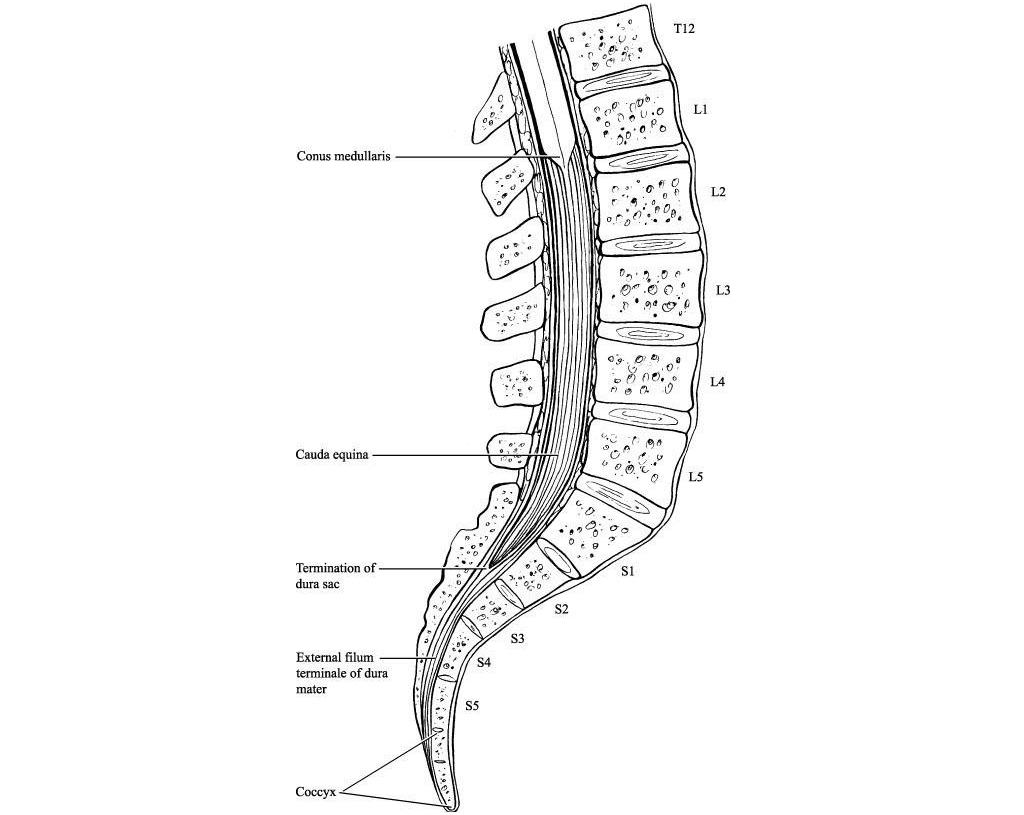
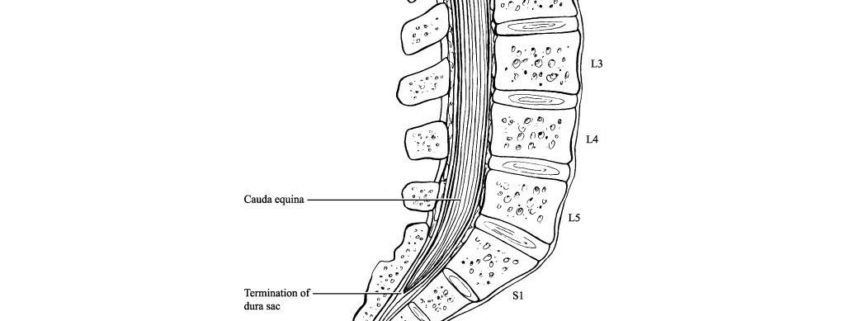
During development, the vertebral column grows more rapidly than the spinal cord. Spinal nerves exit the vertebral column at progressively more oblique angles because of the increasing distance between the spinal cord segments and the corresponding vertebrae. Lumbar and sacral nerves travel nearly vertically down the spinal canal to reach their exiting foramen.
The spinal cord ends at the intervertebral disc between the first and second lumbar vertebrae as a tapered structure called the conus medullaris, consisting of sacral spinal cord segments. The upper border of the conus medullaris is often not well-defined. The fibrous extension of the cord, the filum terminale, is a nonneural element that extends down to the coccyx.
The cauda equina (CE) is a bundle of intradural nerve roots at the end of the spinal cord, in the subarachnoid space distal to the conus medullaris. The CE provides sensory innervation to the saddle area, motor innervation to the sphincters, and parasympathetic innervation to the bladder and lower bowel (i.e., from the left splenic flexure to the rectum).
The nerves in the CE region include lower lumbar and all the sacral nerve roots. The pelvic splanchnic nerves carry preganglionic parasympathetic fibers from S2-S4 to innervate the detrusor muscle of the urinary bladder. Conversely, somatic lower motor neurons from S2-S4 innervate the voluntary muscles of the external anal sphincter and the urethral sphincter via the inferior rectal and the perineal branches of the pudendal nerve, respectively.
Hence, the nerve roots in the CE region carry sensations from the lower extremities, perineal dermatomes, and outgoing motor fibers to the lower extremity myotomes.
The conus medullaris obtains its blood supply primarily from 3-spinal arterial vessels – the anterior median longitudinal arterial trunk and 2-posterolateral trunks. Less prominent sources of blood supply include radicular arterial branches from the aorta, lateral sacral arteries, and the fifth lumbar, iliolumbar, and middle sacral arteries. The latter contributes more to the vascular supply of the cauda equina, although not in a segmental fashion, unlike the blood supply to the peripheral nerves.
The nerve roots may also be supplied by diffusion from the surrounding CSF. Moreover, a proximal area of the nerve roots may have a zone of relative hypo-vascularity.
Pathophysiology
In understanding the pathological basis of any disease involving the conus medullaris, keep in mind that this structure constitutes part of the spinal cord (the distal part of the cord) and is in proximity to the nerve roots. Thus, injuries to this area often yield a combination of upper motor neuron (UMN) and lower motor neuron (LMN) symptoms and signs in the dermatomes and myotomes of the affected segments. On the other hand, a cauda equina lesion is an LMN lesion because the nerve roots are part of the PNS.
Cauda equina syndrome may result from any lesion that compresses CE nerve roots. These nerve roots are particularly susceptible to injury since they have a poorly developed epineurium. A well-developed epineurium, as peripheral nerves have, protects against compressive and tensile stresses.
The microvascular systems of nerve roots have a region of relative hypo-vascularity in their proximal third. Increased vascular permeability and subsequent diffusion from the surrounding cerebral spinal fluid supplement the nutritional supply. This property of increased permeability may be related to the tendency toward edema formation of the nerve roots, which may result in edema compounding initial and sometimes seemingly slight injury.
Etiology
Cauda equina syndrome is caused by any narrowing of the spinal canal that compresses the nerve roots below the level of the spinal cord.
The most common causes of cauda equina and conus medullaris syndromes are the following:
- Lumbar stenosis (multilevel)
- Spinal trauma including fractures
- Herniated nucleus pulposus (cause of 2-6% of cases of cauda equina syndrome)
- Neoplasm, including metastases, astrocytoma, neurofibroma, and meningioma; 20% of all spinal tumors affect this area
- Spinal infection/abscess (e.g., tuberculosis, herpes simplex virus, meningitis, meningovascular syphilis, cytomegalovirus, schistosomiasis)
- Idiopathic (e.g., spinal anesthesia): these syndromes may occur as complications of the procedure or of the anesthetic agent (e.g., hyperbaric lidocaine, tetracaine)
- Spina bifida and subsequent tethered cord syndrome
Other, rare causes include the following:
- Spinal hemorrhage, especially subdural and epidural hemorrhage causing compression within the spinal canal
- Intravascular lymphomatosis
- Congenital anomalies of the spine/filum terminale including tethered cord syndrome
- Conus medullaris lipomas
- Multiple sclerosis
- Spinal arteriovenous malformations
- Late-stage ankylosing spondylitis
- Neurosarcoidosis
- Deep venous thrombosis of the spinal veins (propagated)
- Inferior vena cava thrombosis
A retrospective study of 66 consecutive cases of patients admitted to a neurosurgical unit with suspected cauda equina syndrome found that almost half had no evidence of structural pathology on MRI. These researchers suggested that the symptoms have a functional origin in such cases.
Trauma
Traumatic events leading to fracture or subluxation can lead to compression of the cauda equina. Penetrating trauma can cause damage or compression of the cauda equina. Spinal manipulation resulting in subluxation has caused cauda equina syndrome. Rare cases of sacral insufficiency fractures have been reported to cause cauda equina syndrome. Acute and delayed presentations of CES due to hematomas and posttraumatic arachnoid cysts have also been reported.
Herniated disc
The reported incidence of cauda equina syndrome resulting from herniated lumbar disc varies from 1-15%. Ninety percent of lumbar disc herniations occur either at L4-L5 or L5-S1.
Of cases of herniated discs leading to cauda equina syndrome, 70% occur in patients with a history of chronic low back pain; in 30%, cauda equina syndrome is the first symptom of lumbar disc herniation. Men in the fourth and fifth decades of life are most prone to cauda equina syndrome secondary to disc herniation.
Most cases of cauda equina syndrome secondary to disc herniation involve either a large central disc or an extruded disc fragment that compromises a significant amount of the spinal canal diameter. The presentation may be acute or that of a more protracted course, with the latter bearing a better prognosis. Individuals with congenital stenosis who sustain a disc herniation are more likely to develop cauda equina syndrome because even a small herniation can drastically limit the space available for the nerve roots.
Rare cases of intradural disc herniations have been reported to cause cauda equina syndrome. Myelography, in these instances, typically demonstrates a complete block of the contrast material. If an intradural disc fragment is identified, transdural removal of the extruded disc fragment may be helpful to prevent further stretching of the already compromised nerve root.
Spinal stenosis
Narrowing of the spinal canal can be due to a developmental abnormality or degenerative process. Although unusual, ankylosing spondylitis, spondylosis, and spondylolisthesis have been reported to cause cauda equina syndrome.
Neoplasms
Cauda equina syndrome can be caused by primary or metastatic spinal neoplasms. Primary tumors that can cause CES include myxopapillary ependymoma, schwannoma, and paraganglioma.
Myxopapillary ependymoma is the most common tumor of the filum. Recovery of function after surgery depends on the duration of symptoms and the presence or absence of sphincter dysfunction. Paraganglioma of the filum, when present, needs to be differentiated from other tumors of this region. Although rare, this tumor may present as CES.
Schwannomas are benign encapsulated neoplasms that are structurally identical to a syncytium of Schwann cells. These growths may arise from peripheral or sympathetic nerves. Schwannomas, whether solitary or as a part of a syndrome, may cause CES if present at the level of the conus or filum terminale. Primary tumors that affect the sacrum, such as chordoma and giant cell tumor of the bone, may produce similar symptoms as a result of bony destruction and collapse.
Ependymomas are gliomas derived from relatively undifferentiated ependymal cells. They often originate from the central canal of the spinal cord and tend to be arranged radially around blood vessels. They can lead to increased intracranial pressure (ICP), and cerebrospinal fluid (CSF) has an increased protein level.
Metastatic lesions of the spine are being reported with increasing frequency because of earlier diagnosis, better imaging, and more effective treatment modalities. Although metastasis accounts for most tumors in the spine in general, metastatic tumors in the cauda equina are relatively rare compared with primary tumors.
For the spine in general, sources of spinal metastases are as follows:
- Lung cancer (40-85%)
- Breast cancer (11%)
- Renal cell carcinoma (4%)
- Lymphatic cancer (3%)
- Colorectal cancer (3%)
Although lung cancer is the most common source of spine metastases, in one study, only 0.7% of the lung cancer metastases to the spine produced cauda equina syndrome; most of the metastatic lesions were not at the level of the cauda equina. Up to 8% of patients with prostate cancer experience malignant spinal cord compression; however, the percentage of cases involving cauda equina syndrome is unknown.
The CE region is also a favored site for drop metastases from intracranial ependymoma, germinoma, and other tumors. Genitourinary and gynecologic cancers can metastasize to the conus region, causing neurological compromise.
Inflammatory and infectious conditions
Long-lasting inflammatory conditions of the spine, including Paget disease and ankylosing spondylitis, can lead to cauda equina syndrome secondary to spinal stenosis or fracture.
Infectious conditions, including epidural abscess, can lead to deformity of the nerve roots and spinal cord. Symptoms generally include severe back pain and a rapidly progressing motor weakness.
Infectious causes for cauda equina syndrome may be pyogenic or non-pyogenic. Pyogenic abscesses are generally found in an immunocompromised or poorly nourished host. Staphylococcus aureus causes epidural abscesses in 25-60% of cases, but, recently, an increasing incidence of infections with methicillin-resistant S aureus, Pseudomonas species, and Escherichia coli have been recorded. A high index of suspicion is helpful for correct diagnosis and management.
Non-pyogenic causes for abscess are rare and include tuberculosis. A resurgence of tuberculosis secondary to immunocompromise in individuals with HIV infection requires a high index of suspicion, as the development of cauda equina syndrome may follow an indolent course.
Other uncommon organisms, such as Nocardia asteroides and Streptococcus milleri, have also been reported as a cause of abscess that leads to the development of CES.
Iatrogenic causes
Complications of spinal instrumentation have been reported to cause cases of cauda equina syndrome, including misplaced pedicle screw sand laminar hooks. Continuous spinal anesthesia also has been linked to cases of cauda equina syndrome.
Rare cases of cauda equina syndrome caused by epidural steroid injections, fibrin glue injection, and placement of a free-fat graft have been reported.
Several cases have involved the use of hyperbaric 5% lidocaine for spinal anesthesia. Recommendations are that hyperbaric lidocaine should not be administered in concentrations greater than 2%, with a total dose not to exceed 60 mg.
Medical and surgical situations such as bone screw fixation, fat grafts, lumbar arthrodesis for spondylolisthesis, lumbar discectomy, intradiscal therapy, lumbar puncture forming an epidural hematoma, chiropractic manipulation, and a bolus injection of anesthetic during spinal anesthesia have been related to the development of cauda equina syndrome–like syndromes.
Age-related differences in incidence
Traumatic cauda equina syndrome is not age-specific; atraumatic cauda equina syndrome occurs primarily in adults as a result of surgical morbidity, spinal disc disease, metastatic cancer, or epidural abscess.
History
Patients can present with symptoms of isolated cauda equina syndrome, isolated conus medullaris syndrome, or a combination. The symptoms and signs of cauda equina syndrome tend to be mostly lower motor neuron (LMN) in nature, while that of conus medullaris syndrome are a combination of LMN and upper motor neuron (UMN) effects (see Table 1, below). The history of onset, the duration of symptoms, and the presence of other features or symptoms could point to the possible causes.
Table 1. Symptoms and Signs of Conus Medullaris and Cauda Equina Syndromes (Open Table in a new window)
| Conus Medullaris Syndrome | Cauda Equina Syndrome | |
| Presentation | Sudden and bilateral | Gradual and unilateral |
| Reflexes | Knee jerks preserved but ankle jerks affected | Both ankle and knee jerks affected |
| Radicular pain | Less severe | More severe |
| Low back pain | More | Less |
| Sensory symptoms and signs | Numbness tends to be more localized to perianal area; symmetrical and bilateral; sensory dissociation occurs | Numbness tends to be more localized to saddle area; asymmetrical, maybe unilateral; no sensory dissociation; loss of sensation in specific dermatomes in lower extremities with numbness and paresthesia; possible numbness in pubic area, including glans penis or clitoris |
| Motor strength | Typically, symmetric, hyperreflexic distal paresis of lower limbs that is less marked; fasciculations may be present | Asymmetric areflexic paraplegia that is more marked; fasciculations rare; atrophy more common |
| Impotence | Frequent | Less frequent; erectile dysfunction that includes inability to have erection, inability to maintain erection, lack of sensation in pubic area (including glans penis or clitoris), and inability to ejaculate |
| Sphincter dysfunction | Urinary retention and atonic anal sphincter cause overflow urinary incontinence and fecal incontinence; tend to present early in course of disease | Urinary retention; tends to present late in course of disease |
Symptoms of cauda equina syndrome include the following:
- Low back pain
- Unilateral or bilateral sciatica
- Saddle and perineal hypoesthesia or anesthesia
- Bowel and bladder disturbances
- Lower extremity motor weakness and sensory deficits
- Reduced or absent lower extremity reflexes
Low back pain can be divided into local and radicular pain. Local pain is generally a deep, aching pain resulting from soft-tissue and vertebral body irritation. Radicular pain is generally a sharp, stabbing pain resulting from compression of the dorsal nerve roots. Radicular pain projects in dermatomal distributions. Low back pain in cauda equina syndrome may have some characteristic that suggests something different from the far more common lumbar strain. Patients may report severity or a trigger, such as head-turning, that seems unusual.
Severe pain is an early finding in 96% of patients with cauda equina syndrome secondary to spinal neoplasm. Later findings include lower extremity weakness due to the involvement of the ventral roots. Patients generally develop hypotonia and hyporeflexia. Sensory loss and sphincter dysfunction are also common.
Urinary manifestations of cauda equina syndrome include the following:
- Retention
- Difficulty initiating micturition
- Decreased urethral sensation
- Typically, urinary manifestations begin with urinary retention and are later followed by an overflow urinary incontinence
Bell et al. demonstrated that the accuracy of urinary retention, urinary frequency, urinary incontinence, altered urinary sensation, and altered perineal sensation as indications of possible disc prolapse justified urgent MRI assessment.
Bowel disturbances may include the following:
- Incontinence
- Constipation
- Loss of anal tone and sensation
The initial presentation of bladder/bowel dysfunction may be of difficulty starting or stopping a stream of urine. It may be followed by frank incontinence, first of urine, then of stool. The urinary incontinence is secondary to overflow. It is usually with associated saddle (perineal) anesthesia (the examiner can inquire if toilet paper feels different when the patient wipes).
Physical Examination
The symptoms of cauda equina syndrome are associated with corresponding signs pointing to an LMN or UMN lesion. In addition to the signs listed below, signs of other possible causes should be sought (e.g., an examination of the peripheral pulses to rule out possible vascular cause or ischemia of the conus medullaris).
Pain often is localized to the low back; local tenderness to palpation or percussion may be present. Pain in the legs (or radiating to the legs) is characteristic of cauda equina syndrome. Radicular pain is a common presentation in patients with cauda equina syndrome, usually in association with radicular sensory loss (saddle anesthesia), asymmetric paraplegia with loss of tendon reflexes, muscle atrophy, and bladder dysfunction. The presentation is somewhat similar to and is often confused with conus and epiconus lesions.
Reflex abnormalities may be present; they typically include loss or diminution of reflexes. Hyperactive reflexes may signal spinal cord involvement and exclude the diagnosis of cauda equina syndrome. Sensory abnormalities may be present in the perineal area or lower extremities. Light touch in the perineal area should be tested. Anesthetic areas may show skin breakdown.
Muscle weakness may be present in muscles supplied by affected roots. Muscle wasting may occur in chronic cauda equina syndrome.
Poor anal sphincter tone is characteristic of cauda equina syndrome. Babinski sign or other signs of upper motor neuron involvement suggest a diagnosis other than cauda equina syndrome, possibly spinal cord compression.
In cauda equina syndrome, the peripheral nerve fibers from the sacral segments of the cord, as well as various lumbar dorsal and ventral nerve roots, may also be involved. This results in an asymmetric and higher distribution of motor and sensory symptoms and signs in the lower extremities. The incontinence of bowel and bladder is not severe and develops late for the same reason.
In conus and epiconus lesions, the sacral region neurons (S2-S4) are destroyed. The destruction of these neurons leads to the early and more severe involvement of bowel, urinary bladder, and sexual dysfunction than seen in those with CES. In contrast, for the same reason, the motor and sensory symptoms in the lower extremities are often not very severe, and only the distal parts of the limb musculature are involved.
The anatomical proximity of the conus medullaris, the epiconus, and the cauda equina can lead to 2 of these anatomical structures being involved via a single lesion, resulting in an overlap of symptomatology.
The salient features and findings of cauda equina syndrome and conus medullaris syndrome are listed in Table 4, below.
Cauda equina syndrome
In cauda equina syndrome, muscle strength in the lower extremities is diminished. This may be specific to the involved nerve roots as listed below, with the lower lumbar and sacral roots more affected, leading to diminished strength in the glutei muscles, hamstring muscles (i.e., semimembranosus, semitendinosus, biceps femoris), and the gastrocnemius and soleus muscles.
The sensation is decreased to pinprick and light touch in a dermatomal pattern corresponding to the affected nerve roots. This includes saddle anesthesia (sometimes including the glans penis or clitoris) and decreased sensation in the lower extremities in the distribution of lumbar and sacral nerves. Vibration sense may also be affected. The sensation of the glans penis or clitoris should be examined.
Muscle stretch reflexes may be absent or diminished in the corresponding nerve roots. Babinski reflex is diminished or absent.
Bulbocavernosus reflexes may be absent or diminished; this should always be tested.
Anal sphincter tone is patulous and should always be tested since it can define the completeness of the injury (with bulbocavernosus reflex); it is also useful in monitoring recovery from the injury.
Urinary incontinence could also occur secondary to loss of urinary sphincter tone; this may also present initially as urinary retention secondary to a flaccid bladder.
Muscle tone in the lower extremities is decreased, which is consistent with an LMN lesion.
Conus medullaris syndrome
Patients may exhibit hypertonicity, especially if the lesion is isolated and primarily UMN.
Signs are almost identical to those of the cauda equina syndrome, except that in conus medullaris syndrome signs are more likely to be bilateral; sacral segments occasionally show preserved bulbocavernosus reflexes and normal or increased anal sphincter tone; the muscle stretch reflex may be hyperreflexic, especially if the conus medullaris syndrome (i.e., UMN lesion) is isolated; Babinski reflex may affect the extensors; and muscle tone might be increased (i.e., spasticity).
Other signs include papilledema (rare, occurs in lower spinal cord tumors), cutaneous abnormalities (e.g., cutaneous angioma, pilonidal sinus that may be present in dermoid or epidermoid tumors), distended bladder due to areflexia, and other spinal abnormalities (noted on lower back examination) predisposing the patient to the syndrome.
Muscle strength
Physical examination for cauda equina or conus medullaris syndromes would be incomplete without tests for the sensation of the saddle and perineal areas, bulbocavernosus reflex, cremasteric reflex, and anal sphincter tone, findings for all of which are likely to be abnormal.
Muscle strength of the following muscles should be tested to determine the level of lesion:
- L2 – Hip flexors (iliopsoas)
- L3 – Knee extensors (quadriceps)
- L4 – Ankle dorsiflexors (tibialis anterior)
- L5 – Big toe extensors (extensor hallucis longus)
- S1 – Ankle plantar flexors (gastrocnemius/soleus)
ASIA impairment scale
In defining impairments associated with a spinal cord lesion, the American Spinal Cord Injury Association (ASIA) impairment scale is used in determining the level and extent of the injury.
This scale should also be used in defining the extent of conus medullaris syndrome/cauda equina syndrome. The scale is as follows:
- A – Complete; no sensory or motor function preserved in sacral segments S4-S5
- B – Incomplete; sensory, but not motor, function preserved below the neurologic level and extends through sacral segments S4-S5
- C – Incomplete; motor function preserved below the neurologic level, and most key muscles below the neurologic level have a muscle grade less than 3
- D – Incomplete; motor function preserved below the neurologic level, and most key muscles below the neurologic level have a muscle grade greater than or equal to 3
- E – Normal; sensory and motor function normal
The injury should be described using this scale, for example, ASIA class A. Most patients with cauda equina/conus medullaris syndrome are in ASIA class A or B initially and gradually improve to class C, D, or E.
Complications
Complications include the following:
- Thromboembolic phenomena
- Neurogenic bladder/bowel
- Erectile dysfunction
- Pressure ulcers
- Heterotopic ossification
- Osteoporosis
- Chronic neuropathic pain
- Spasticity/contractures
- Recurrent urinary tract infections
- Urethral stricture
- Bladder calculi
- Depression
Diagnostic Considerations
Conus medullaris infarction should be considered in the differential diagnosis, and a source of emboli should be sought by ultrasound to rule out an abdominal aortic aneurysm. Heterotopic ossification should be ruled out by a triple-bone scan in a patient with pain and swelling of the lower extremity in whom deep venous thrombosis (DVT) has been ruled out. In other words, heterotopic ossification should always be considered as a differential diagnosis of DVT in these patients.
Metastatic malignant neoplasms of the spine should be ruled out, and the primary source sought as part of the workup in any patient presenting with any of the symptoms listed in Clinical.
Other problems to be considered include the following:
- Amyloidosis with deposits in the spinal cord
- Ankylosing spondylitis and other spondyloarthropathy
- Charcot-Marie-Tooth disease (types 1 and 3)
- Intravascular lymphomatosis
- Lipomas within the spine
- Lumbar stenosis (multilevel)
- Paget disease of the spine
- Spinal infection/abscess and meningitis
- Tethered cord syndrome/short filum terminale
- Vascular intermittent claudication
Differential Diagnoses
- Acute Inflammatory Demyelinating Polyradiculoneuropathy
- Amyotrophic Lateral Sclerosis in Physical Medicine and Rehabilitation
- Diabetic Neuropathy
- Guillain-Barré Syndrome
- Multiple Sclerosis
- Neoplasms, Spinal Cord
- Neuromuscular and Myopathic Complications of HIV
- Neurosarcoidosis
- Spinal Cord Infections
- Traumatic Peripheral Nerve Lesions
Approach Considerations
The diagnosis of cauda equina syndrome generally can be made by medical history and physical examination findings. Radiologic and laboratory studies are used to confirm the diagnosis and for localizing the site of the pathology and the underlying cause.
Myelography, computed tomography, and MRI are used in specific cases, with a high degree of accuracy. Each test can be used to determine the level of pathology and aid in the determination of the cause of the syndrome. Bone scans may detect malignant tumors or metastases and inflammatory conditions affecting the vertebrae.
Due to its ability to depict the soft tissues, MRI generally has been the favored imaging study for assisting the physician in the diagnosis of cauda equina syndrome. Urgent MRI is recommended for all patients who have new-onset urinary symptoms with associated back pain or sciatica.
Nevertheless, the superiority of MRI over CT is only suggested by case reports. Early consultation with the appropriate subspecialty is encouraged to guide imaging studies.
Depending on the findings from the history and physical examination, laboratory studies can include basic blood tests, chemistries, fasting blood sugar, sedimentation rate, syphilis, and Lyme serologies. CSF examination should also be included if signs of meningitis are present.
Alteration in bladder function may be assessed empirically by obtaining urine via catheterization. A significant volume with little or no urge to void, or as a post-void residual, may indicate bladder dysfunction. Bedside ultrasonography may be used to estimate or measure post-void residual bladder volume.
Urodynamic studies are useful to evaluate the degree and the cause of sphincter dysfunction, as well as to monitor the recovery of bladder function following decompression surgery. Intraoperative monitoring of somatosensory and motor evoked potentials allows for the evaluation of radiculopathy and neuropathy.
Blood Studies
The following studies may help to define possible causes and any associated pathology, especially other causes of lesions in the lower spinal cord or cauda equina:
- CBC count, blood glucose, electrolytes, blood urea nitrogen (BUN), and creatinine – As part of the workup to rule out associated anemia, infection, and renal dysfunction, especially in associated retroperitoneal mass
- Erythrocyte sedimentation rate (ESR) – Elevation may point to an inflammatory pathology
- Syphilis serology to rule out meningovascular syphilis
Radiography
Plain radiography is unlikely to be helpful in cauda equina syndrome but may be helpful in cases of traumatic injury, in search of destructive changes, disc-space narrowing, or spondylolysis. For example, plain radiographs of the lumbosacral spine may depict early changes in vertebral erosions secondary to tumors and spina bifida.
Chest radiography is indicated to rule out a pulmonary source of pathology that could affect the lumbosacral spine (e.g., malignant tumor, tuberculosis). Follow-up chest CT may be required.
Magnetic Resonance Imaging
MRI with gadolinium contrast of the lumbosacral area is the diagnostic test of choice to define pathology in the areas of the conus medullaris and cauda equina. It provides a complete radiographic assessment of the spine than other tests; plain x-rays and CT scans may be normal. Gadolinium contrast MRI also may be able to rule out abdominal aneurysm, which could be the source of emboli causing conus medullaris infarction.
Schwannomas are visible using myelography, but MRI is the criterion standard. Schwannomas are isointense on T1 images, hyperintense on T2 images, and enhanced with gadolinium contrast. With infectious conditions, MRI may display the abnormal appearance of the nerve roots being forced to one side of the dural sac.
Other Tests and Procedures
Needle electromyography (EMG)may show evidence of acute denervation, especially in cauda equina lesions and multilevel lumbar spinal stenosis. EMG studies also could help in predicting prognosis and monitoring recovery. Performing needle EMG of the bilateral external anal sphincter muscles is recommended.
Nerve conduction studies, especially of the pudendal nerve, may rule out more distal peripheral nerve lesions.
Somatosensory evoked potentials (SSEPs)could be done as part of the workup to rule out multiple sclerosis, which could present initially as a lower spinal cord syndrome.
Duplex ultrasound of peripheral vessels may rule out compromised vasculature as a possible cause of associated claudication.
Lumbar puncture should be performed to examine the CSF to rule out an inflammatory disease of the meninges or spinal cord.
Management
Specific treatment is directed at the primary cause. The general treatment goals are to minimize the extent of an injury and to treat ensuing general complications.
Other medical treatment options are useful in certain patients, depending upon the underlying cause of the cauda equina syndrome. Anti-inflammatory agents and steroids can be effective in patients with inflammatory processes, including ankylosing spondylitis.
Patients with cauda equina syndrome secondary to infectious causes should receive appropriate antibiotic therapy. Patients with spinal neoplasms should be evaluated for the suitability of chemotherapy and radiation therapy.
Methylprednisolone should be administered within 8-hours of injury. No evidence exists of any benefit if it is started more than 8 hours after injury; on the contrary, late treatment may have detrimental effects.
For penetrating trauma, steroids have not shown significant benefits.
Administration of ganglioside GM1 sodium salt beginning within 72- hours of injury may be beneficial; the dose is 100 mg IV QD for 18-32 days.
Tirilazad mesylate (a nonglucocorticoid 21-aminosteroid) has been proven to be of benefit in animals and is currently under investigation. It inhibits lipid peroxidation and hydrolysis in the same manner as glucocorticoids.
Caution should be used in all forms of medical management for cauda equina syndrome. Any patient with true cauda equina syndrome with symptoms of saddle anesthesia and/or bilateral lower extremity weakness or loss of bowel or bladder control should undergo no more than 24-hours of initial medical management. If no relief of symptoms is achieved during this period, immediate surgical decompression is necessary to minimize the chances of permanent neurologic injury.
Emergency Department Care
No proven medical treatment exists. Treatment generally is directed at the underlying cause of cauda equina syndrome.
Surgery is controversial. The timing of decompression is controversial, with immediate, early, and late surgical decompression showing varying results. For mechanical compression of the cauda due to disc herniation, surgical intervention may be indicated.
Admit patients to the appropriate service (usually neurology, neurosurgery, or orthopedic surgery) with frequent neurologic checks. Ideally, the admitting physician or service should examine the patient at the time of admission. Patients in whom acute cauda equina syndrome is being considered should not be treated or investigated on an outpatient basis without evaluation by a consultant and/or appropriate imaging.
Surgical Decompression
In acute compression of the conus medullaris or cauda equina, surgical decompression as soon as possible becomes mandatory. The goal is to relieve the pressure on the nerves of the cauda equina by removing the compressing agent and increasing the space in the spinal canal. Traditionally, cauda equina syndrome has been considered a surgical emergency, with surgical decompression considered necessary within 48 hours after the onset of symptoms, and preferably within 6-h of injury if the patient is medically stable and able to undergo the procedure.
A study of the outcome of surgery for intradural CE and conus medullaris tumors reported overall neurologic improvement in 62% of patients. The reversibility of preoperative neurologic deficits was found to be related to the interval between symptom onset and the timing of the surgery. Ependymoma and carcinoma metastases were associated with higher morbidity. The findings suggested that early surgery is advisable because more than 94% of patients maintain at least their preoperative status, and more than 60% improve during follow-up.
In 14-patients with CES presenting with low back pain, sciatica, and impaired sphincteric function, surgery resulted in relief of back or leg pain and sensory improvement in all patients; full return of bladder and bowel control in 12-patients; and improvement in lower extremity strength in 9-patients. The surgical procedures were all performed 1-3 months after the onset of sphincteric dysfunction.
A study of 33-patients with CES from lumbar disc herniation found that operation within 24-hours led to significant improvements in motor strength.
A large study that evaluated the timing of surgery for CES between 2000 and 2014 confirmed that the performance of decompression within 0 or 1 day after patient admission is associated with improved in-patient outcomes, including lower complication and mortality rates.
For patients in whom a herniated disc is the cause of cauda equina syndrome, a laminotomy or laminectomy for decompression of the canal is recommended, followed by gentle retraction and discectomy.
In a chronic presentation with less severe symptoms, decompression could be delayed to optimize the patient’s medical condition; with this precaution, decompression is less likely to lead to irreversible neurological damage.
Surgery is safe in pregnant patients; however, obstetric support is advisable in case of complications to the fetus.
The intraoperative use of somatosensory and motor-evoked potentials (SSEPs) may be helpful in monitoring the patient.
Surgical treatment may include laminectomy and instrumentation/fusionfor stabilization or discectomy. After spinal surgery, internal stabilization with fixation devices may be needed in the same operative setting or at a later date.
Prognosis
Morbidity, and especially mortality rates, are determined by the underlying etiology. Multiple conditions can result in cauda equina or conus medullaris syndrome. The prognosis improves if a definitive cause is identified, and appropriate treatment occurs early in the course. Surgical decompression may be performed emergently, or, in some patients, delayed, depending on the etiology. Residual weakness, incontinence, impotence, and/or sensory abnormalities are potential problems if therapy is delayed.
Investigators have attempted to identify specific criteria that can aid in predicting the prognosis of patients with cauda equina syndrome. Patients with bilateral sciatica have been reported to have a less favorable prognosis than patients with unilateral pain. Patients with complete perineal anesthesia are more likely to have permanent paralysis of the bladder.
The extent of perineal or saddle sensory deficit has been reported to be the most important predictor of recovery. Patients with unilateral deficits have a better prognosis than patients with bilateral deficits. Females and patients with bowel dysfunction have been reported to have worse outcomes postoperatively.
Prognosis can be predicted with the American Spinal Injury Association (ASIA) impairment scale (see Physical Examination), as follows:
- ASIA A: 90% of patients remain incapable of functional ambulation (reciprocal gait of 200 feet or more)
- ASIA B: 72% of patients are unable to attain functional ambulation
- ASIA C/D: 13% are unable to attain functional ambulation 1 year after injury
Ambulatory motor index also is used to predict ambulatory capability. It is calculated by scoring hip flexion, hip abduction, hip extension, knee extension, and knee flexion on both sides, using a 4-point scale (0=absent, 1=trace/poor, 2=fair, 3=good or normal); the score is expressed as a percentage of the maximum score of 30. Prognostic significance is as follows:
- A patient with a score of 60% or more has a good chance for community ambulation with no more than one knee-ankle-foot orthosis (KAFO)
- A patient with a score of 79% or higher may not need an orthosis
- A patient with a score of 40% or less may require 2 KAFOs for community ambulation
Patient Education
Patient education needs will vary with the type and severity of persistent deficits, and may include the following:
- Training in self-catheterization and finger fecal disimpaction, if required
- Use of measures to prevent pressure ulcers, such as skin inspection/care, positioning, turning and transferring techniques, use of skin protectors, or pressure-reducing support surfaces
- Maintenance of endurance and strength-training exercises
- Regular follow-up by the consulting teams who treated the patient in the hospital
- Instructions on how and when medications should be taken and when follow-up laboratory tests should be performed